Kobayashi Group
Please click here for new Web site of Kobayashi laboratory
Previous studies
2000-2005 (Under direction of Prof. Masahiro Hirama in Tohoku Univ.)
(1) Study of nine-membered enediyne antibiotics: Total synthesis and mode of action
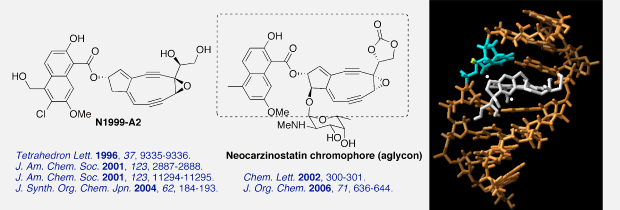
N1999-A2 is the latest member of the highly strained nine-membered enediyne antibiotic family. In 2001, we achieved first total synthesis of N1999-A2 and elucidated the stereochemistry of the natural product. We also provided the chemical evidence of a thiol-triggered aromatization and showed the unique DNA cleavage profiles of a series of stereoisomers, which proved that the stereochemical orientation of the C11-naphthoate unit played a dominant role in defining efficiency and specificity in the DNA recognition and cleavage process. In 2006, we succeeded in the synthesis of neocarzinostatin chromophore aglycon, which is an active component of SMANCS, a chemotherapeutic agent against primary hepatocellular carcinoma.
(2) Synthetic study of ciguatoxins, the causative toxins of ciguatera poisoning
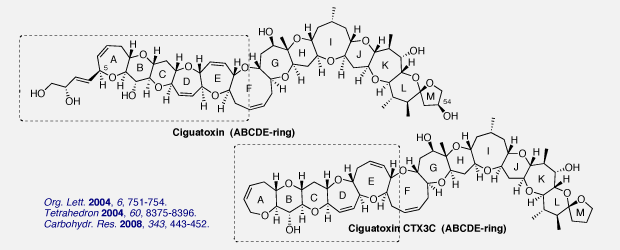
Ciguatoxins (CTXs), the causative toxins of ciguatera seafood poisoning, are huge polycyclic ethers composed of thirteen ether rings ranging from 5- to 9-membered. We succeeded in the syntheses of the ABCDE ring moiety of ciguatoxin and CTX3C. The syntheses feature a short-step installation of the dihydroxybutenyl substituent into the ABCD ring (for ciguatoxin) and the efficient construction of the E ring using the O,S-acetal forming reaction (for ciguatoxin) or the diastereoselective Aldol reaction (for CTX3C).
2005-2009
(3) Synthetic study of polycyclic natural products from mushroom (with Prof. Masahiro Toyota in Osaka Prefecture University)
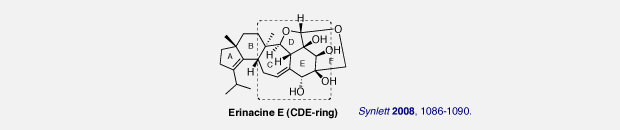
Erinacine E is a potent stimulator of nerve growth factor synthesis (NGF-synthesis).
We have developed an efficient route to the CDE ring system of erinacine E featuring the substrate-controlled stereoselective Grignard addition, Pummerer-type cyclization, and RCM.
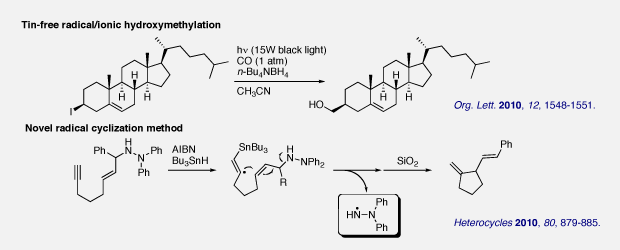
(4) Investigation of new radical reactions (Under direction of Prof. Ilhyong Ryu in Osaka Prefecture Univ.)
In the course of our program to develop the new radical reaction with CO, tin-free radical/ionic hydroxymethylation reaction was investigated. The reaction proceeded efficiently in the presence of tetrabutylammonium borohydride as the hydrogen source under atmospheric pressure of CO with photoirradiation. We also developed a new radical cyclization method employing hydrazine as the leaving group.
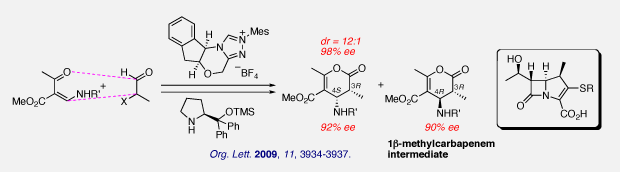
(5) Practical synthesis of a key synthetic intermediate of 1β-methylcarbapenems
We have achieved an enantioselective synthesis of 3,4,5,6-tetrasubstituted 3,4-dihydropyran-2-ones bearing a nitrogen substituent at C4 by means of organocatalytic cycloadditions. This is the first example using the organocatalytic reaction to construct the four contiguous asymmetric centers of 1β-methylcarbapenem skeletons.